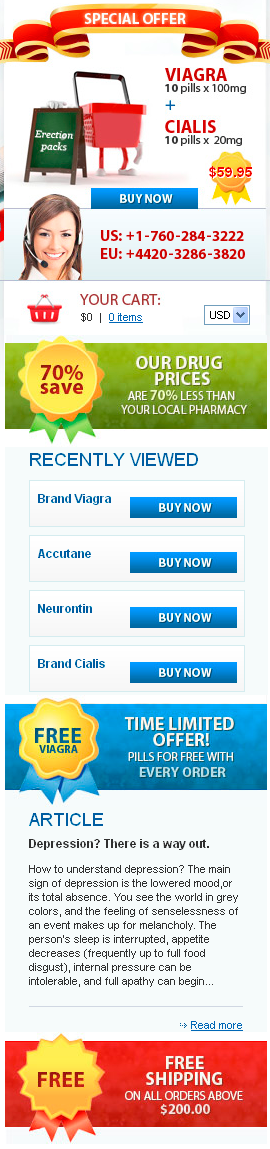
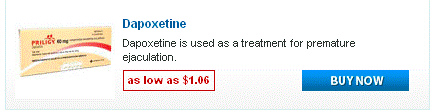
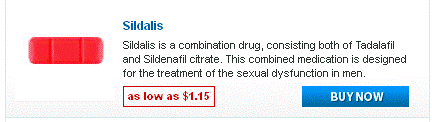
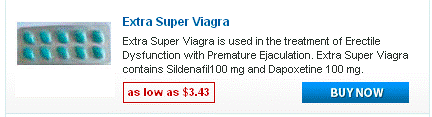
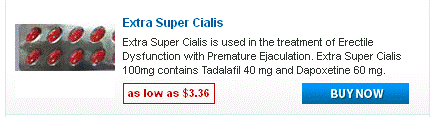
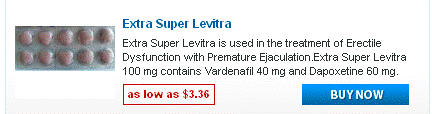
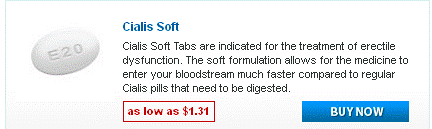
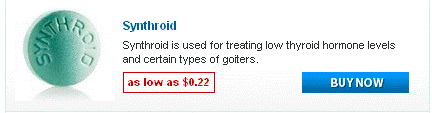
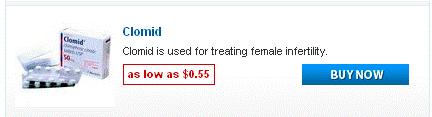
Doxycycline monohydrate boasts a tetracycline core structure, featuring a four-ring system with various hydroxyl and amino groups. This specific arrangement grants it its unique antimicrobial properties. The monohydrate designation indicates the presence of a single water molecule bound to each doxycycline molecule. This hydration affects its solubility, making it readily dissolvable in slightly acidic environments. Its molecular formula is C22H24N2O8·H2O, and its molar mass is approximately 444.4 g/mol.
Solubility and Stability
Doxycycline monohydrate exhibits varying solubility depending on the pH. It readily dissolves in dilute mineral acids but is less soluble in water and organic solvents. Light exposure degrades doxycycline, therefore, storing it in amber glass containers protects its stability. The substance is stable in its solid form when kept in dry conditions away from light. Decomposition can occur under extreme temperatures or in the presence of strong bases. Careful handling and storage ensures the prolonged efficacy of the medication. Precise solubility data is available in various pharmacopeias.